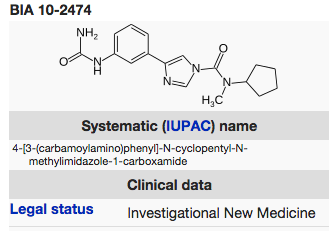
In mid-January we ran this item about a French clinical trial of a FAAH-inhibitor drug that proved fatal to one participant and injurious to five others who required hospitalization. The drug was produced by Bial, a Portuguese company and given a code name, BIA 10-2474. The ‘first-in-human’ phase I trial was undertaken by a company called Biotrial in Rennes.
Some basic facts about the trial were reported in Nature January 21:
• The trial recruited 128 healthy volunteers aged 18–55, who were paid €1,900 (US$2,060) each.• The trial had tested escalating single doses of the drug without observing any serious adverse side effects.
• The six participants who fell ill were the first to receive repeat higher doses over the course of several days.
• The first participant to fall ill experienced adverse symptoms on 10 January and died on 17 January.
• Biotrial halted the trial on 11 January; the other five affected people were hospitalized in the days that followed.• One of these patients has since been discharged, and the condition of the other four is judged to be serious but stable.• Authorities are contacting the 84 other people who received the drug at lower doses to arrange medical check-ups; none of the 18 given neurological check-ups over the weekend showed any of the symptoms seen in the hospitalized people.
“Many researchers believe that BIA 10-2474 is acting ‘off target’,” according to the piece in Nature, “inhibiting a protein other than an FAAH. To investigate, researchers could radioactively label the compound and test it on brain tissue from cadavers to ‘fish out’ the proteins it binds to.”
Nobody ever points out that the development of FAAH inhibitors (and MAGL-inhibitors) was and is a costly, risky alternative to letting people use cannabis as medicine. BIA 10-2474, according to Nature, was “aimed at treating anxiety and motor disorders associated with Parkinson’s disease, and chronic pain in people with cancer and other conditions.” Conditions treatable by ingesting cannabinoids.
Cannabinoid researchers rely on funding from NIDA and they have used it to elucidate the workings of the endocannabinoid system. It’s the decision-makers at NIDA who channel research into areas that are only fruitful inadvertently.
Thanks to Alec Dixon of SC labs for forwarding the story in Nature.