From the Food & Drug Administration:
The FDA is investigating a rise in reports of serious adverse events involving Limbrel. While a range of adverse events have been reported, two serious and potentially life-threatening medical conditions are among them: drug-induced liver injury and hypersensitivity pneumonitis. In total, the FDA has received 200 adverse event reports regarding Limbrel; 30 of these contained sufficient information to use the Council for International Organizations of Medical Sciences (CIOMS) causality assessment method to determine the likelihood that an association between the consumption of Limbrel and the adverse events reported exists.
On November 8, 2017, the FDA first contacted Primus Pharmaceuticals regarding the adverse events and requested information about the formula for Limbrel. The formula is being reviewed by the FDA. The FDA also obtained product samples from the company and the samples are undergoing testing.
On November 30, 2017, the FDA reiterated, to Primus Pharmaceuticals, the agency’s safety concerns and serious health risks associated with continued use of the product. The FDA recommended a voluntary recall, but the company declined to take the product off the market.
On December 18, 2017, the FDA formally requested that Primus Pharmaceuticals recall all non-expired lots of Limbrel products, because of the risk of liver injury and other serious adverse events associated with the products. Specifically, the FDA requested that Primus Pharmaceuticals recall Limbrel (flavocoxid) 250 mg capsules, Limbrel250 (250 mg flavocoxid with 50 mg citrated zinc bisglycinate) capsules, Limbrel (flavocoxid) 500 mg capsules and Limbrel500 (500 mg flavocoxid with 50 mg citrated zinc bisglycinate).
The FDA continues to investigate Primus Pharmaceuticals and the manufacturing process for Limbrel, and will share updates as they become available.
A lawyer on the prowl for Limbrel victims posted this:
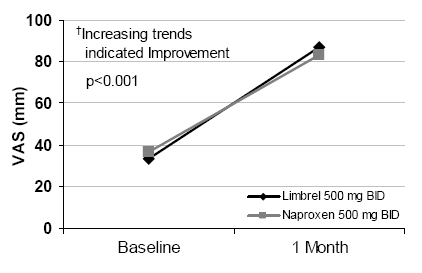
Limbrel was tested in a placebo-controlled double-blind study of more than 200 patients that took place over a 12-week period. An open-label study of 1,067 subjects that took 60 days was also conducted. However, this was a relatively small number of individuals examined under the controlled conditions. Additionally, a long-term follow-up was not conducted.
From the new O’Shaughnessy’s: FDA reversals.
When I was researching the marketing of Prozac, a reliable source at FDA estimated that the number of adverse event reports received after the release of any given drug represented “probably about two percent and definitely less than 10 percent” of those actually taking place. The patient has to tell the doctor who has to find time to fill out the paperwork for FDA. —FG